
by msander | Dec 1, 2014 | Gr 9-10 Science, Gr. 11-12 Chemistry
Scientific discoveries don’t always result from methodically conducted experiments. In fact, several have resulted purely by accident. This process is sometimes called serendipity. True story – one of my professors described serendipity as “reaching into a...

by msander | Nov 27, 2014 | Gr 4-6 Science & Tech, Gr. 11-12 Chemistry, Gr. 11-12 Physics
Mention the word magnetism and most of us think of one iron-based object attracting another. This form of magnetism is called ferromagnetism. Iron, nickel and cobalt are the most common elements that exhibit ferromagnetic properties. However, two other less...

by msander | Nov 13, 2014 | Gr 9-10 Science, Gr. 11-12 Chemistry, Gr. 11-12 Physics
As Carl Sagan famously said, “We are made of star stuff.” Whoa. It’s a mind-boggling thought, but what exactly did he mean? Check out the video to find out… Thanks to the American Association of Chemistry Teachers for making this video...
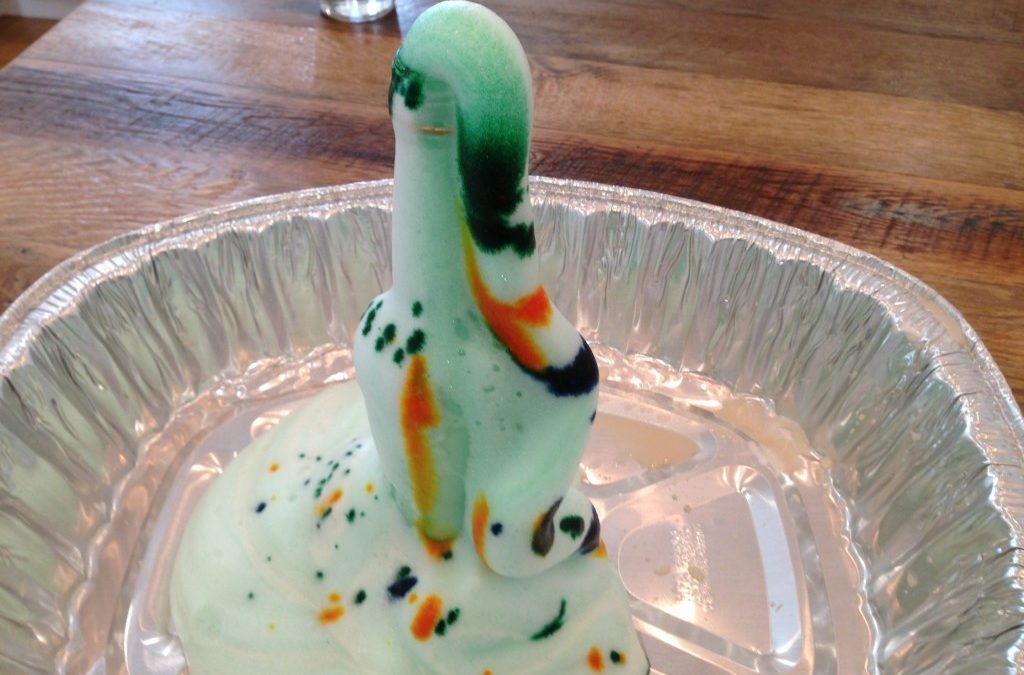
by msander | Nov 13, 2014 | Gr 9-10 Science, Gr. 11-12 Chemistry, Safety
Elephant Toothpaste is a popular demonstration to introduce the concept of decomposition reactions. The chemical reaction involved is the decomposition of hydrogen peroxide into oxygen gas and water. The reaction is very slow at room temperature....
by cbrookallred | Nov 3, 2014 | All Science Teachers, Gr. 11-12 Chemistry, Teaching Strategies
>>> Written by Nick Key… Through the first 8 years of my teaching career, I felt a growing level of apathy in my students. I needed a new way to engage them (and me). So, over the past three years, I have been part of a growing movement of educators...

by cbrookallred | Oct 20, 2014 | Gr 9-10 Science, Gr. 11-12 Chemistry, Safety
Many teachers demonstrate the reaction of lithium, sodium and potassium with water when discussing the trend in reactivity of the alkali metals. In order to conduct this demo safely, it is imperative that only a small quantity of metal is used and that appropriate...




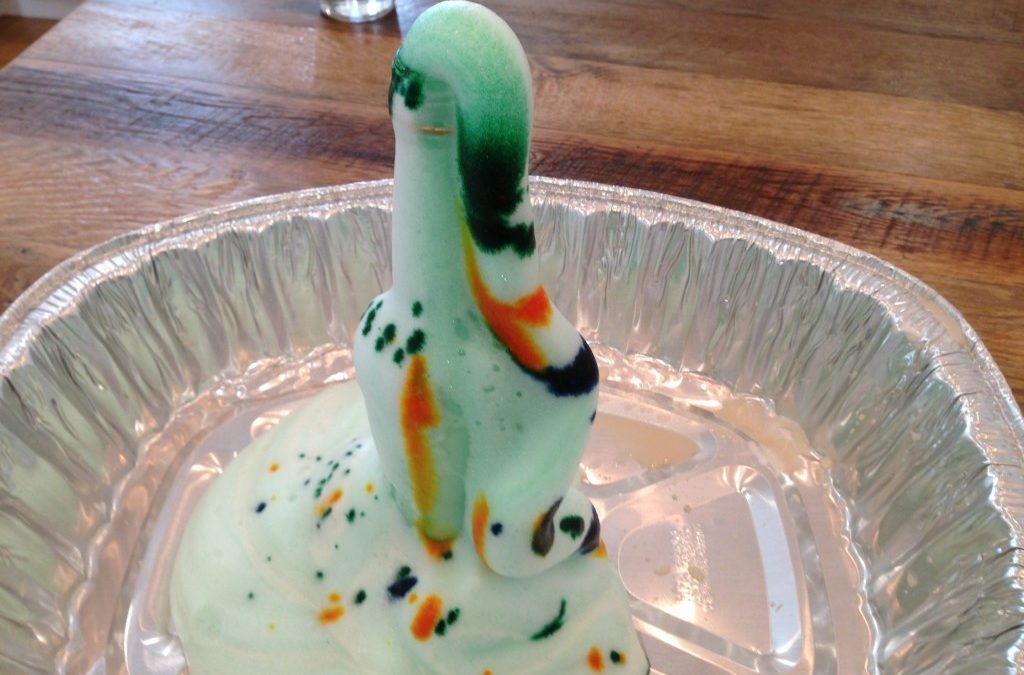



Recent Comments